Interview with Dr. Deborah Knapp on Screening Scottish Terriers
Dr. Deborah Knapp, the senior author of the study, was interviewed and shared the key features & findings of the study below. You can read the full published results on PubMed.
What makes this study unique?
This study represents what we hope will be the first step in a major paradigm shift in cancer management. Far too often and across many cancer types in veterinary and human medicine, the cancer is not found until it is already advanced in the organ where it starts, i.e. at the “local disease level”, or the cancer has already spread to other organs. The physician or veterinarian is left “reacting” to the cancer and playing “catch up” in cancer therapy. Exceptions to this in humans would include the treatment of early cancer found through colonoscopy, mammography, or PSA screening. But most other cancer types in humans and most cancers in dogs are found “late”, when therapies are less effective.
Our study was different in that our goal was to find the cancer, in this case bladder cancer, at an early state through screening dogs with no clinical signs, i.e. no “symptoms” of the cancer. To our knowledge this was the first published cancer screening and early detection study in dogs. We focused on dogs that have a very high bladder risk based on their breed, Scottish Terriers.
Why is screening and early treatment expected to be a better approach?

There are several things about early cancer that should make it more responsive to treatment.
- At the early stages of cancer, the dog’s anti-tumor immune responses are more effective at combatting the cancer. In contrast, in more advanced cancer, the immune system has become exhausted and is ineffective in controlling the cancer.
- The blood flow to the tumor is better in early cancer allowing for good cancer drug delivery, whereas in more advanced cancer, the blood only reaches part of the cancer mass(es).
- There are less active molecular pathways for drug resistance in early cancer, whereas as cancer persist in the body, it acquires many ways to avoid being killed by drugs.
- Dogs with early cancer are in better overall health, allowing them to handle treatment well.
- If dogs do not have any cancer-related symptoms at early diagnosis, then even if the tumor is just kept stable, i.e. not necessarily shrinking but not growing, then the dogs will continue to be symptom free.
Why did you focus on Scottish Terriers?
There were two broad reasons to focus on Scotties. First, since Scotties have such a high inherited risk of bladder cancer, there is a great need to help these dogs. Second, by focusing on Scotties, crucial early detection and early intervention studies can be successfully completed with far fewer dogs. If we were evaluating dogs across all breeds, we would need to enroll hundreds if not thousands of dogs to find early cancer, because across all breeds the cancer is not that common. Whereas if we focus on Scotties we expect to find early cancer by studying a few hundred dogs or less. In other words, with Scotties, we know where to look.
What were the main goals of the study?
The study was designed to answer 4 crucial questions:
- Is it possible to find early cancer prior to any “symptoms”, called “clinical signs” in dogs, or bladder dysfunction in the Scotties?
- Does finding the cancer early actually do any good? Does it lead to better outcomes?
- How do the features of the cancer including molecular characteristics differ between the early cancer detected by screening and later cancer detected in dogs that already have symptoms and bladder dysfunction? In what ways does the canine cancer resemble human bladder cancer?
- Are there other tests that might be considered for screening beyond the ultrasound and urine sediment exam used in this screening study?
How did you find enough Scotties for the study?
This was a phenomenal “team” effort involving the Purdue team ( Figure 2), many veterinarians in the field, and very importantly the Scottish Terrier community. The Scottie community was phenomenal! The Scottish Terrier Club of America provided a lot of the funding needed to conduct the study. People like Dr. Marcia Dawson, a veterinarian and champion of the Scottie breed, local breed clubs, and others in the Scottie community helped spread the word, recruited dogs to participate, and held fund raising events such as the Door County Scottie Rally in Wisconsin and other events. The study would not have happened without that support. It is worth pointing that there were certainly skeptics that thought we would not find enough dogs and that we would not find early cancer. But this amazing team made it happen!

Did all of the dogs come to Purdue for the study?

No, we enrolled dogs at three different locations. We held screening clinics at the Purdue University Veterinary Hospital throughout the year ( Figure 3). Then the Purdue team went on the road to two other practices twice a year. Dr. Byron McCall and Ms. Lisa Hills organized screening clinics at the Capitol Illini Veterinary Hospital in Springfield IL, and Drs. Daley and Grahler, and Rose and Jack Shacklett organized screening clinics at the Metropolitan Veterinary Specialty Hospital in Louisville Kentucky. And Scottie owners drove from multiple states to allow their dogs to participate. All this support was key!
How did you do the screening?
We recruited Scotties who were at least 6 years old and had no outward evidence of any bladder disease. We performed a very detailed bladder ultrasound exam following a standardized protocol to detect irregularities or masses in the bladder lining, urethra, or prostate; a physical exam including a rectal exam to detect abnormalities in the urethra prostate, or lymph nodes in the area; and urinalysis with urine sediment exam to detect abnormal bladder lining cells ( Figure 4). We scheduled this every 6 months for 3 years. If these screening test became positive or suspicious, we performed cystoscopy and biopsy. In some cases we made diagnoses at necropsy.

How did the study turn out? What were some of the key findings?
The results from the study actually far exceeded our expectations!
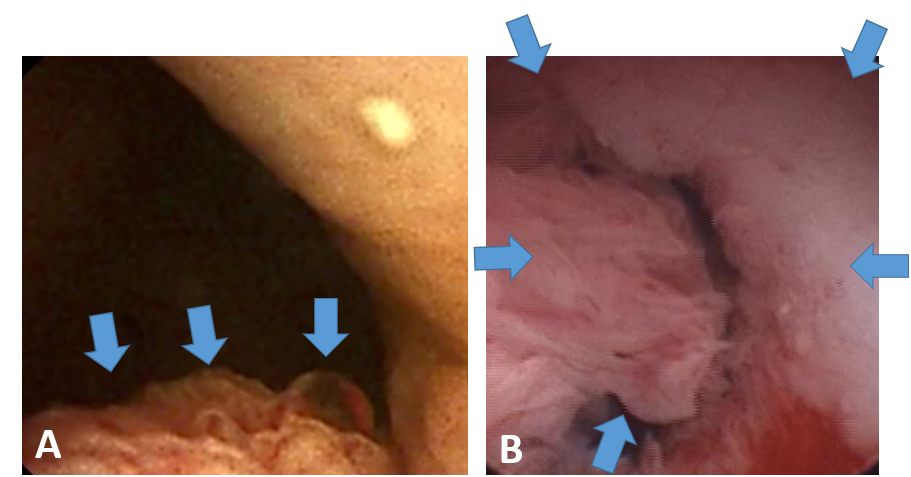
First, we did, in fact, find bladder cancer early! We found early cancer in 32 of 120 dogs (27%) in the three years of screening, and this was when the dogs had no outward evidence of cancer. These dogs appeared normal to their owners. The tumors were usually quite small (see Figure 5).
Second, finding the cancer early did lead to better outcomes. In the Scotties with early cancer detected via screening, the dogs were allowed to participate in a treatment trial of a drug called deracoxib (Dermaxx TM, Elanco, Greenfield, IN). This treatment is a simple, reasonably priced, conservative, oral treatment that is usually well tolerated in dogs. The complete plus partial remission rate with deracoxib was 42%! To understand why the 42% remission rate stands out, we need to consider the remission rate of deracoxib and similar drugs in dogs that come to see us when they have symptoms and “later” more typically-advanced bladder cancer. In multiple published studies, the remission rate is only around 20% in the dogs with later cancer (Table 1). While the cancer was not cured in most of the dogs, it was well controlled with good quality of life for a median of 300 days. Essentially, the cancer was managed like a chronic medical condition while the dogs continued to enjoy life.
| Treatment | Number of dogs | Remission rate (%)* | Median PFI (days) | Median Survival (days) |
|---|---|---|---|---|
| Deracoxib in early bladder cancer found through screening, current study |
24 |
42% |
304 |
615 |
| Deracoxib in previous study in dogs with “symptomatic” more advanced bladder cancer |
26 |
17% |
133 |
323 |
| Piroxicam, another drug in this class, in dogs with “symptomatic” more advanced bladder cancer |
76 |
21% |
120 |
244 |
*In these analyses, we focused most on remission rate because PFI and survival could potentially be affected by lead-time bias.
People are already asking what would happen if we treated the dogs with more intensive therapy. We do not know. It could possibly lead to better outcomes. But, it could also lead to more treatment-related side effects. More studies are needed!
What did your team find related to the molecular analyses?
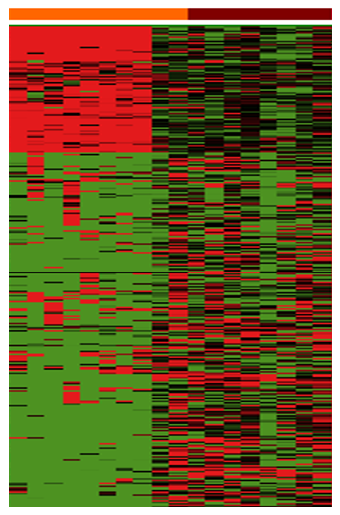
The molecular findings were really fascinating. We looked at patterns in RNA-sequencing data to see the expression of genes in the tumors. The findings were particularly interesting in the dogs that have what we refer to as “luminal molecular subtype tumors”. In these dogs, the early cancer was much less complex, and had activation of fewer molecular pathways that would cause drug resistance and cancer progression. The differences were quite dramatic ( Figure 6). On the other hand, dogs that have what we refer to as “basal molecular subtype tumors” had aggressive cancer features from the “get go”. The reason this is important is that it opens up doors to figure out at the molecular level how cancer develops, how it worsens, and how we can intervene to stop the process. We also confirmed that the cancer in Scotties is very similar to high grade invasive bladder cancer in people. Our work is always aimed at helping pet dogs and helping people facing cancer, and this study is no exception.
You mentioned other tests. Did you identify other tests that appear useful in bladder cancer screening in dogs?
The short answer is “no”, but this was one of the more informative aspects of the study, although not in the way the veterinary community would have predicted! We evaluated two tests that can be done on urine to try to detect bladder cancer. At each visit, we had to first prioritize the use of the urine from each dog for the urinalysis and urine sediment exam, the main endpoints in our study. We then used leftover sample for these two additional urine tests, and analyzed the results at the end of the study when we knew which dogs had or had not developed bladder cancer.
Veterinary Bladder Tumor Antigen (V-BTA) Test
The first extra urine test was a commercially available kit called the Veterinary Bladder Tumor Antigen (V-BTA) Test. This test has been limited by false positive results in dogs with blood in the urine from any cause whether cancer is present or not. We thought it might work better in the early disease setting when there was less blood in the urine. But, this ended up not being the case.
We performed the V-BTA test on 434 urine samples from 118 dogs. As is the case with more advanced cancer, false positives were common. In many dogs, the test results were positive when cancer was not present and did not develop. The positive predictive value of the test was 33%. This means for the dogs with a positive test, that cancer was present in 33% of the dogs, but cancer was not present in 67% of the dogs. The negative predictive value was 80%. This means for 100 dogs with a negative test, 80 dogs did not have cancer, but 20 dogs did have cancer.
BRAF Mutation Test
An approach that has gained attention for screening dogs for bladder cancer in recent years has been to analyze urine samples from the dogs to detect a mutation in the BRAF gene, specifically the BRAF V595E mutation, which is the dog homologue of human BRAF V600E found in many types of human cancer. Work from multiple groups has indicated that the BRAF V595E mutation is present in 80% or more of canine bladder cancer. Studies to determine the actual predictive value of this mutation for the presence or emergence of canine TCC, however, have been very limited, and very little has been published in the peer-reviewed literature. Our screening study provided an excellent opportunity to determine the value of this test. The results were very unexpected, although vitally important.
With collaborators S. Tersey and J. Nelson at the University of Chicago, we used a droplet digital PCR assay to detect the BRAF V595E mutation in 383 urine sediment samples available from 117 dogs. The BRAF V595E mutation was found in the majority of dogs with high grade cancer, in 4 of 5 dogs with lower grade cancer, in 2 of 3 dogs with a preneoplastic condition called dysplasia, and in 3 dogs that later developed TCC. The presence of the mutation was common as expected, although it did not distinguish between the typical high grade cancer and the less common less aggressive low grade tumors that would be treated differently. The results that were perhaps most striking however, were that the BRAF V595E mutation was detected in the urine of 33 of 80 dogs that did not have cancer and that did not develop detectable cancer over a median (“average”) followup time of 18 month. Some of these dogs remained free of detectable cancer for 5 years after the positive BRAF test.
What does this mean for using BRAF mutation testing to detect and diagnose bladder cancer in dogs?
That is a very good question. We analyzed our data to determine the “predictive value” of BRAF testing in our study. With the many false positive results (i.e. presence of the mutation in the absence of cancer), the positive predictive value of the presence of the BRAF mutation in predicting the presence of bladder cancer was only 44%. This means that for every 100 dogs with a positive BRAF test, that 44 dogs had cancer and 56 dogs did not have cancer. The negative predictive value was better at 92%. This means for every 100 dogs with a negative test, that 92 dogs did not have cancer, and that 8 dogs did have cancer, i.e. were missed by the test. We noticed that several dogs had a “low mutation fraction” meaning the mutation was only present in a few “alleles” of the gene. When we considered the low fraction as a “negative” result, then the positive predictive value increased to 66%, but still not where it needs to be for a screening or diagnostic test, and the negative predictive value was slightly worse in that analysis.
We also asked if using the results from multiple tests together would work better in predicting the presence of bladder cancer. We asked if the presence of the BRAF mutation, abnormal ultrasound findings, and the presence of atypical epithelial cells in the urine altogether would line up with a diagnosis of TCC. Interestingly, it did not. Some of the dogs had ultrasound changes of a thick irregular bladder wall or bladder masses, abnormal cells in urine, and a positive BRAF test, but did not have cancer. This included one of the dogs with ulcerative cystitis found on cystoscopic biopsy, a dog with a paraprostatic sarcoma confirmed at necropsy, and dogs that had an apex nodule that may represent an anatomic variant in the breed. We do not necessarily think these other conditions “harbor” a BRAF mutation. While this will require further study, we think it is possible that there are some dogs that have cells in the urinary tract that have acquired the BRAF mutation, but that those cells do not survive or that they do not grow and form a tumor. Work is ongoing to better understand this.
Another important point to make related to BRAF testing is that when the test was repeated at multiple time points in dogs undergoing treatment, there was no association between the presence of the mutation, nor the “mutation fraction” (percent of gene alleles that had the mutation) and the response to deracoxib. We also observed this lack of correlation in an earlier study with a drug called vemurafenib. In both studies, we found that the mutation fraction at diagnosis did not predict the response to treatment. And, the change in the BRAF V595E mutation during treatment did not correlate with the change in tumor size either. This is not entirely surprising. A cancer mass is made up of many parts with different clusters of cells. We do not know which of those clusters have the BRAF mutation, and we do not know which clusters are killed with therapy. This means we still need to carefully measure bladder tumors in a standardized and consistent way over time to determine how well therapies are working.
Does this mean you will move away from using a BRAF test for detecting bladder cancer?
Well, there have been multiple groups including ours, that have been waiting on more studies to understand how well BRAF testing works before using the test. For us, it’s not moving away from the BRAF test because we have not been using it. “Thinking out loud”, if we were to do a BRAF test and it was positive for the presence of the mutation, then we would still do an ultrasound and urinalysis with sediment exam, and biopsy if those tests were positive. If we were to do the BRAF test and it was negative, we would still do an ultrasound and urine sediment exam and follow with cystoscopy and biopsy if those tests were suspicious.
Is there a “positive side” to the poor predictive value of the BRAF mutation testing for bladder cancer detection?
Yes, I can think of at least two upsides to this finding.
First, we now have a naturally-occurring “model system” to study the factors that lead to the acquisition of the BRAF mutation, AND the factors that then drive the development of cancer from there, but perhaps more importantly, the factors that keep some dogs from developing cancer even in the presence of this “cancer driver mutation”. The human homologue of the BRAF mutation that we study in dogs, i.e. human BRAF V600E , is an important driver in 8% of all human cancer across cancer types. The mutation is not inherited, it is acquired in the life of the human or dog. If we can learn why some individuals acquire the mutation and other do not, and how to derail the cancer development after this mutation is acquired, that will be huge in developing strategies to save human and canine lives. One of important parts of this ongoing research is to determine the role of the immune system in attacking tumors cells that have BRAF mutations.
A second upside to the findings would be to serve as an important reminder to all of us in the veterinary profession and pet owning community. We must demand rigorous testing of any new product presented to us, whether that be a new drug, supplements, genetic testing, or other cancer detection tests. Since the process to bring products like this to the market and to launch website and social media post about the products is not monitored or regulated, we must step up and demand more. We must insist on rigorous testing and publication of findings in the peer-reviewed literature before using new products.
There is still one scenario in which BRAF testing could have a role in managing dog bladder cancer. In a trial published by our group in 2021, the drug, vemurafenib, which targets tumor cells with the BRAF mutation, had very good antitumor activity in dogs with bladder cancer. It is expected that this beneficial effect will be for dogs that have the BRAF mutation in their cancer. While vemurafenib is not affordable, as other drugs in that class become available and affordable, there could be a role to use BRAF testing to select dogs for such therapies.
What suggestions would you give pet owners that cannot pursue scoping?
We hear this question a lot. We know there are tens of thousands of dogs that develop bladder cancer each year, and that many pet owners cannot afford everything that veterinarians might suggest. One option is to look for clinical trials that may be open in your area because these often come with some funds to help pay for the dog’s care, while helping others. There is another approach that the pet owner could discuss with their veterinarian. If bladder cancer is suspected based on clinical signs (“symptoms”) or imaging or urinalysis or combinations of this, and a urinary tract infection is not the cause, then one option is to treat the dog with a drug in the deracoxib class, i.e. nonsteroidal anti-inflammatory drugs, “NSAIDs” also called cyclooxygenase inhibitors or COX inhibitors. These drugs could be helpful if it turns out that cancer is present or if an inflammatory condition is present in the absence of cancer. These drugs could help the dog feel better, and the risk of side effects is not particularly high. It is still important to watch for side effects though such as loss of appetite, unexplained vomiting, and dark black tarry-looking stools, and stop the drug should this happen, and then talk to your veterinarian about treatment choices from then on.
What is next? And, do you recommend screening dogs in all high risk breeds for bladder cancer?
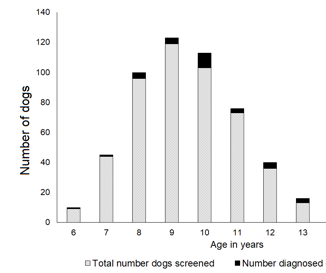
This study has focused on Scotties. While we expect screening to be advantageous to other dogs in high risk breeds such as West Highland White Terriers, Shetland Sheepdogs, Beagles and some others, this has not been specifically studied yet. A point to consider is that Scotties have a 20X higher risk for bladder cancer, and these other breeds have a 3-5X higher risk for bladder cancer. This makes us anticipate that we will need to screen 4X more Westies than Scotties to find the same number of dogs with cancer (Figure 7). Having seen the benefits of screening though, there is certainly interest in screening dogs in high risk breeds beyond Scotties.
You also asked what is next. We are continuing to analyze other types of data from this study. By being well integrated into the Purdue University Center for Cancer Research, we have had the opportunity to work with scientists to analyze metabolomics data and genomic data. This is ongoing. We are also looking at environment exposure data to determine what factors might contribute to bladder cancer risk. Please stay tuned!
Dr. Knapp, thank you for your team’s amazing work and for answering these questions. We invite pet owners and veterinarians interested in the study to read the full article at PubMed.

