
Purdue University is continuing to lend expertise and resources to help fight the COVID-19 pandemic by increasing Indiana’s testing capacity through the capabilities of the Animal Disease Diagnostic Laboratory (ADDL) at the Purdue University College of Veterinary Medicine. Thanks to a collaborative effort, the ADDL is utilizing its skills and expertise in the detection of pathogens to conduct COVID-19 tests on specimens from partner hospitals with the goal of providing results the same day the samples are received.
The ADDL began COVID-19 testing after obtaining the Clinical Laboratory Improvement Amendments (CLIA) certification required for laboratories performing testing on human samples. The lab was certified in a matter of days after Purdue leaders suggested using the ADDL to conduct tests and address the state’s limited testing capacity and need for resources. State Health Commissioner Kris Box, M.D., FACOG, said in a recent daily briefing with Gov. Eric Holcomb that resources are still limited for COVID-19 testing in Indiana as the number of cases continue to rise.
“The College of Veterinary Medicine has a long history of providing services to protect animal and human health,” said Purdue Veterinary Medicine Dean Willie Reed. “Providing COVID-19 testing is yet another way to support the citizens of Indiana during this unprecedented public health crisis. The project was shepherded by David Broecker, chief innovation and collaboration officer for the Purdue Research Foundation, and involves several partners including the foundation, the Indiana State Department of Health, Parkview Hospital, and the Purdue community.”
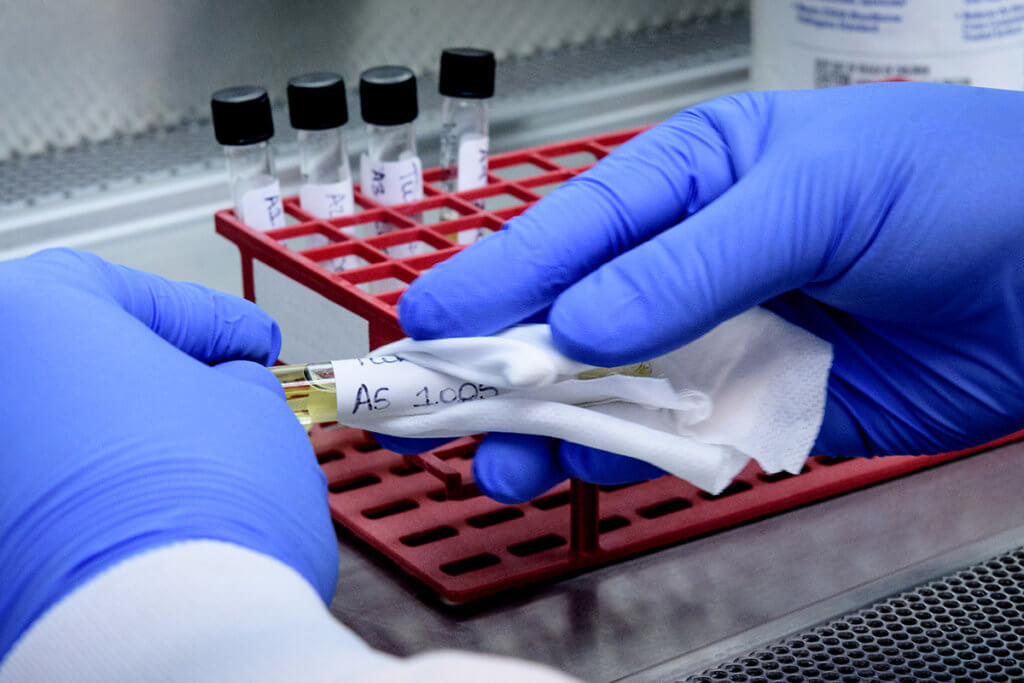
ADDL Director Kenitra Hendrix says the lab had a unique opportunity to assist with COVID-19 testing in Indiana because of its expertise in providing infectious disease testing and immunity surveillance for animals across the state. Dr. Hendrix explains the human samples are being tested using a PCR (Polymerase Chain Reaction) testing method that the ADDL uses regularly for animal specimens. “Our plan to help our state in this way does not diminish our capacity for animal testing,” Dr. Hendrix said. “Our primary mission is diagnostic support of animal health and the safety of the food supply, and that will remain our first priority.”
The ADDL team, working with state health authorities, adjusted its intake and sample processing procedures to gain the necessary certification to use the PCR testing capability on human specimens. “Our team has worked diligently to prepare to perform this testing,” Dr. Hendrix said. “It is rewarding to be able to take on this role in support of the citizens of our state.”
The plan is to expand testing in collaboration with other hospitals, as ADDL testing capacity allows, with all samples sent directly from partner hospitals to the ADDL. To avoid a testing backlog, hospitals interested in working with the ADDL are required to complete the “Partnership Inquiry” form.
The project is a cross-disciplinary, collaborative effort with the Indiana State Department of Health, the Purdue Research Foundation and the Purdue community. “Indiana continues to increase our capabilities and preparedness for rapidly testing patients to support our front-line health care workers,” Dr. Box said. “We are grateful to Purdue and the ADDL for helping to make testing more available for Hoosiers who are helping others during this pandemic.”
Fort Wayne-based Parkview Health provided clinical expertise on obtaining a CLIA license and setting up the lab for human testing. “It still takes several days to get results that are sent out to central labs across the country,” said Dr. Michael J. Mirro, chief academic research officer at Parkview Health. “Even the new tests are limited by supply constraints. What Purdue has done is fantastic and shows the ingenuity associated with creative problem solving. Paired with the significant amount of time invested by the Parkview lab team, we believe this will have a positive impact on the state’s testing capacity.”
The ADDL is a Biosafety Level 2 (BSL-2) facility accredited by the American Association of Veterinary Laboratory Diagnosticians (AAVLD). BSL-2 laboratories are used to study moderate-risk infectious agents or toxins. Dr. Hendrix says that additional partnerships with other hospitals across the state are possible. More information and the form for potential partners to complete if they wish to be contacted should testing capacity increase, are available at https://purdue.vet/covid19testing. Click here to view a complete news release about the ADDL COVID-19 testing program.
To make a contribution in support of Purdue’s COVID-19 testing facility and the Animal Disease Diagnostic Laboratory, click here and see CORONAVIRUS (COVID-19) TESTING LAB.

